Components of An Electric Vehicle Battery
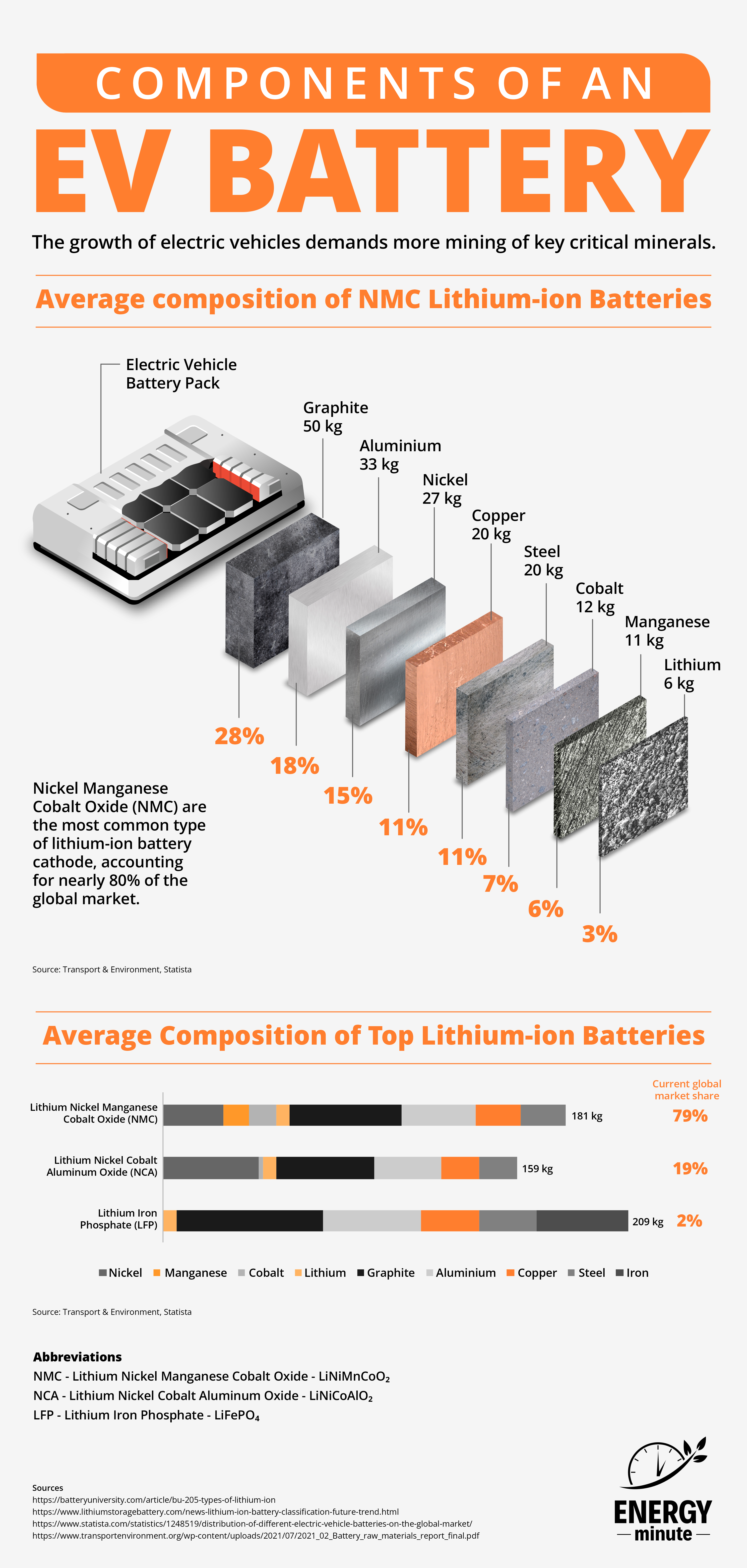
An electric vehicle battery is typically made up of several components, including:
- Electrodes: The positive and negative electrodes are the heart of the battery. They are usually made of a metal oxide for the positive electrode and graphite for the negative electrode.
- Separator: The separator is a thin layer that separates the positive and negative electrodes in the battery. It is typically made of a porous material that allows the flow of ions while preventing electrical contact between the electrodes.
- Electrolyte: The electrolyte is a liquid or gel substance that allows the flow of ions between the positive and negative electrodes. It is typically a lithium salt in a solvent such as ethylene carbonate.
- Current collectors: The current collectors are metal plates that collect the electrical current produced by the electrodes. They are usually made of aluminum or copper.
- Casing: The casing is the outer shell of the battery that protects it from damage and contains the electrolyte. It is typically made of plastic or metal.
- Thermal management system: The thermal management system helps regulate the temperature of the battery to prevent overheating or freezing, which can affect its performance and lifespan. It may include cooling or heating elements, as well as sensors and controls to monitor and adjust the temperature.
All of these components work together to produce and store electrical energy in an electric vehicle battery.
Improving performance of lithium-ion batteries
Lithium-ion batteries have undergone significant changes and improvements over the years, and they continue to evolve. Here are a few ways in which lithium-ion batteries are changing:
- Higher Energy Density: Energy density refers to the amount of energy that can be stored per unit of volume or mass. Over time, researchers have been able to increase the energy density of lithium-ion batteries, which means they can store more energy in the same amount of space or weight. This has led to the development of smaller and lighter batteries with longer run times.
- Longer Lifespan: One of the major drawbacks of lithium-ion batteries has been their limited lifespan, but improvements in the manufacturing process and changes in the chemical composition of the batteries have led to longer lifespans. This means that lithium-ion batteries can be used for a longer period of time before needing to be replaced.
- Faster Charging: Another area of improvement for lithium-ion batteries is charging time. Faster charging means less downtime for devices and more convenience for users. Some manufacturers have introduced fast-charging technology that allows batteries to be charged in a matter of minutes rather than hours.
- Safety Features: Lithium-ion batteries can be prone to overheating and catching fire, but researchers and manufacturers have developed new safety features to prevent these incidents. For example, some batteries now have built-in temperature sensors that can detect when the battery is getting too hot and shut it down to prevent damage or fires.
- Recycling: As the use of lithium-ion batteries continues to grow, there is increasing concern about the environmental impact of disposing of them. To address this issue, researchers and manufacturers are developing new methods of recycling lithium-ion batteries, which can help to reduce waste and preserve valuable materials.
Overall, lithium-ion batteries continue to evolve and improve, and these advancements have significant implications for a wide range of industries, from consumer electronics to renewable energy storage.
Abbreviations
NMC – Lithium Nickel Manganese Cobalt Oxide – LiNiMnCoO 2
NCA – Lithium Nickel Cobalt Aluminum Oxide – LiNiCoAlO 2
LFP – Lithium Iron Phosphate – LiFePO 4
Sources
https://batteryuniversity.com/article/bu-205-types-of-lithium-ion
https://www.lithiumstoragebattery.com/news-lithium-ion-battery-classification-future-trend.html




