Carbon Intensities of Hydrogen
About Hydrogen
Hydrogen is the most abundant element in the universe, but it is not commonly found on Earth in its elemental form. It is typically bound to other elements, such as in water or hydrocarbons. Hydrogen has a wide range of potential uses, including fuel for transportation, power generation, and industrial processes.
Making Hydrogen
There are several ways to produce hydrogen, including electrolysis, steam methane reforming, and biomass gasification.
- Electrolysis involves using an electric current to split water into hydrogen and oxygen.
- Steam methane reforming involves reacting natural gas with steam to produce hydrogen and carbon monoxide, which can be further processed into hydrogen.
- Biomass gasification involves heating organic matter in the absence of oxygen to produce a gas that contains hydrogen and other gases.
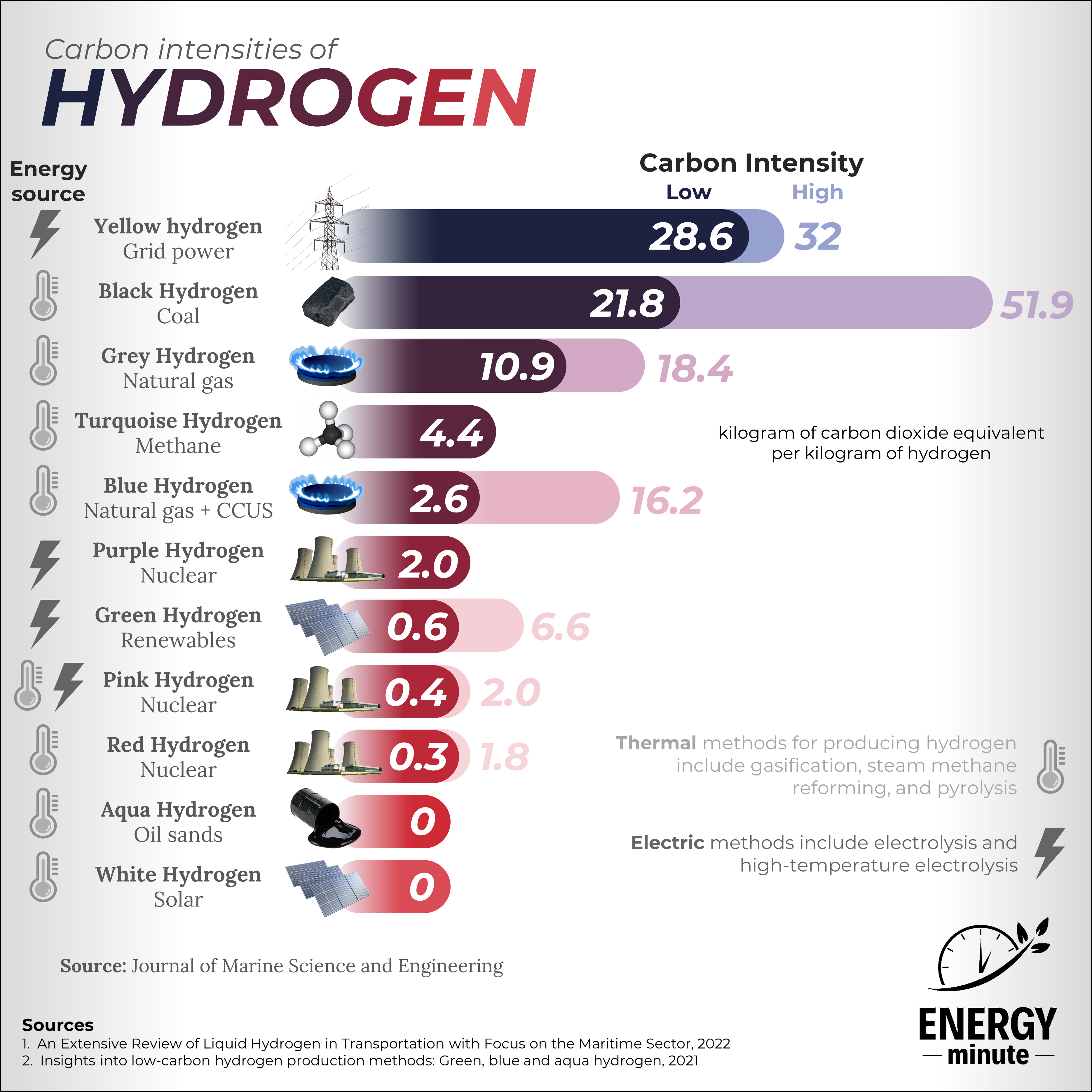
Hydrogen can be produced using many different methods with multiple feedstocks. The most common today include grey and brown hydrogen, which heat up both natural gas and coal to produce hydrogen with a high carbon intensity. Companies are looking to expand low-emission hydrogen, particularly green and blue hydrogen.
Thermal methods for producing hydrogen include gasification, steam methane reforming, and pyrolysis.
Electric methods include electrolysis and high-temperature electrolysis.
Hydrogen as a Fuel
Hydrogen has the potential to be a key component of a low-carbon energy system, as it produces no emissions when used as a fuel. Another way hydrogen can help address climate change is by reducing emissions from the transportation sector. Hydrogen fuel cell vehicles are already on the market and can provide a zero-emission alternative to traditional gasoline or diesel vehicles. Fuel cell vehicles use hydrogen to produce electricity, which powers an electric motor to propel the vehicle. The only emission from a fuel cell vehicle is water vapor, making them a clean alternative to traditional vehicles.
Hydrogen for Power
Hydrogen can also be used in power generation. Fuel cells can be used to generate electricity in stationary applications, such as for backup power or to provide electricity to remote areas. In addition, hydrogen can be burned in gas turbines to produce electricity. When burned in a turbine, hydrogen produces no greenhouse gas emissions and can be used to generate power in a way that is more environmentally friendly than traditional fossil fuels.
Hydrogen for Industry
In industry, hydrogen can be used as a feedstock to produce chemicals, such as ammonia and methanol. Hydrogen can also be used to reduce emissions in industrial processes, such as steel production. By using hydrogen instead of coal to reduce iron oxide to iron in a blast furnace, the emissions associated with steel production can be significantly reduced.
Future Hurdles
One challenge to the widespread adoption of hydrogen is the cost of producing and transporting it. Currently, the majority of hydrogen is produced using fossil fuels, which means that it is not a completely clean energy source. However, as renewable energy sources become more prevalent and the cost of producing green hydrogen decreases, it is expected that the use of hydrogen as a clean energy source will become more widespread.
Another challenge is the infrastructure needed to support the use of hydrogen as a fuel. Fueling stations for hydrogen fuel cell vehicles are still relatively rare, which can limit the adoption of this technology. However, as more fuel cell vehicles are sold, it is expected that the infrastructure to support them will also grow.
In summary, hydrogen has the potential to be an important part of a low-carbon energy system. It can be produced using renewable energy sources and has a wide range of potential uses, including as a fuel for transportation, power generation, and industrial processes. While there are still challenges to overcome, the increasing interest in hydrogen as a clean energy source suggests that it will play an important role in addressing climate change in the future.
Sources:
An Extensive Review of Liquid Hydrogen in Transportation with Focus on the Maritime Sector, 2022
Insights into low-carbon hydrogen production methods: Green, blue and aqua hydrogen, 2021




